Biomechanical alignment is routinely assessed through visual observation of joint position and body segment orientation. However, this positional paradigm fails to account for the dynamic force-regulation mechanisms that govern musculoskeletal system behaviour under progressive load. A mechanistically grounded reconceptualisation of alignment — as an emergent property of force-vector management, torque distribution, and centre-of-mass stability — is both theoretically justified and clinically necessary.
To present the MMSX Alignment Spectrum, classifying alignment into five operationally defined grades (A through E) based on load-dependent changes in moment arm geometry, force vector trajectories, torque distribution patterns, and proximity to tissue tolerance thresholds; and to delineate the clinical significance of the Grade C-to-D transition.
Narrative synthesis of peer-reviewed biomechanical, orthopaedic, sports science, and motor control literature. Evidence drawn from EMG studies, inverse dynamics analyses, motion capture research, tissue tolerance modelling, and in vivo loading investigations.
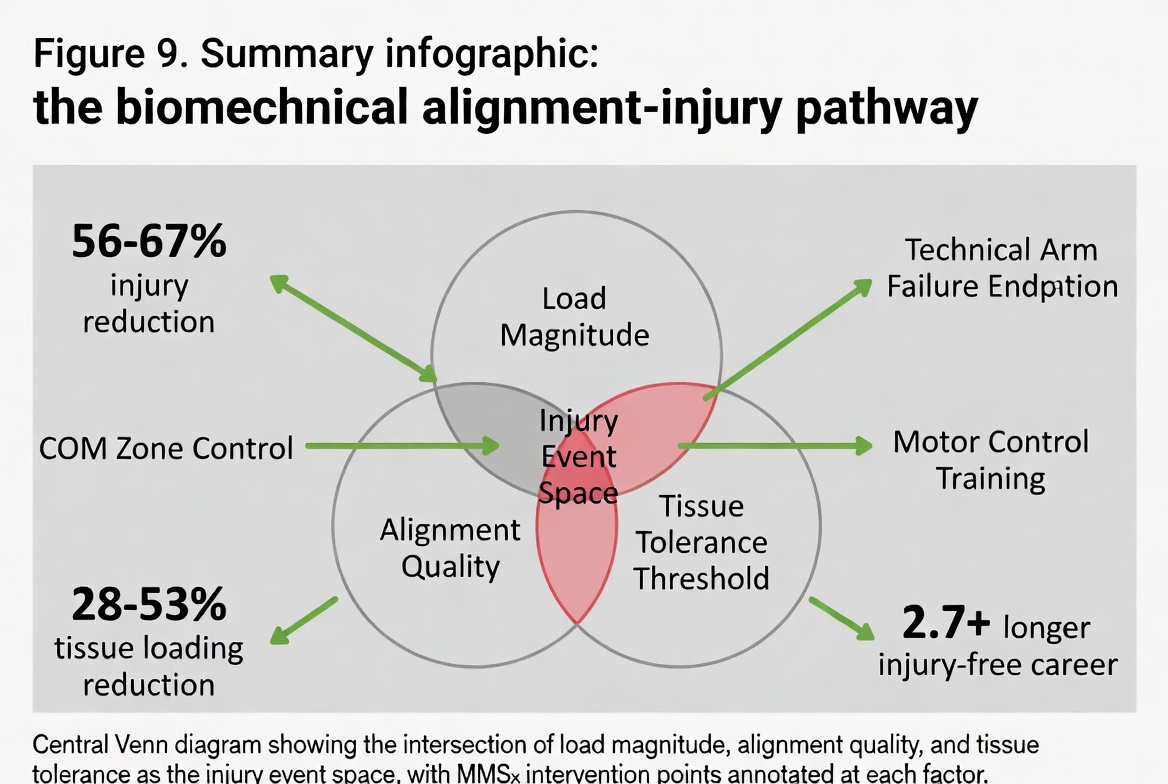
Alignment is load-dependent and continuously variable. The Grade C-to-D transition represents a critical inflection point unidentifiable through positional observation alone. Grade D is characterised by elevated injury risk and diminished mechanical efficiency; Grade E constitutes frank structural failure risk.
Alignment is a continuous, load-dependent force-regulation outcome. The MMSX Alignment Spectrum provides a scalable, evidence-congruent instrument with significant implications for injury prevention, load management, and performance optimisation.
El alineamiento biomecánico se evalúa habitualmente mediante la observación visual de la posición articular. Sin embargo, este paradigma posicional no considera los mecanismos dinámicos de regulación de fuerzas bajo carga progresiva.
Presentar el Espectro de Alineamiento MMSX, clasificando el alineamiento en cinco grados operacionalmente definidos (A–E) basados en cambios dependientes de la carga en la geometría del brazo de momento, trayectorias de vectores de fuerza y proximidad a los umbrales de tolerancia tisular.
Síntesis narrativa de literatura biomecánica revisada por pares, incluyendo estudios de EMG, análisis de dinámica inversa, investigación de captura de movimiento y modelado de tolerancia tisular.
El alineamiento es dependiente de la carga. La transición de Grado C a D representa un punto de inflexión crítico no identificable únicamente mediante observación posicional.
El alineamiento es un resultado continuo de regulación de fuerzas. El Espectro MMSX proporciona un instrumento escalable y congruente con la evidencia para la prevención de lesiones y la optimización del rendimiento.
يُقيَّم التوافق البيوميكانيكي عادةً من خلال الملاحظة البصرية لموضع المفصل. غير أن هذا النموذج الوضعي لا يراعي آليات تنظيم القوى الديناميكية.
تقديم طيف التوافق MMSX، الذي يصنّف التوافق ضمن خمس درجات معرَّفة تشغيلياً استناداً إلى التغيرات المعتمدة على الحمل في هندسة ذراع العزم ومسارات متجهات القوى.
تجميع سردي للأدبيات البيوميكانيكية المحكّمة، مستمدًّا من دراسات EMG وتحليل الديناميكا العكسية ونمذجة تحمّل الأنسجة.
يتّسم التوافق بالتبعية للحمل. يمثّل الانتقال من الدرجة C إلى D نقطة تحوّل حرجة لا يمكن تحديدها عبر الملاحظة الوضعية وحدها.
التوافق نتيجة مستمرة لتنظيم القوى. يوفر طيف MMSX أداةً قابلةً للتوسع ومتوافقةً مع الأدلة للوقاية من الإصابات وتحسين الأداء.
Introduction
The concept of biomechanical alignment occupies a foundational position in sports science, clinical rehabilitation, strength and conditioning, and movement-based medicine. Despite its central importance, the dominant operational definition of alignment in practice remains stubbornly positional — evaluated through observation of joint angles, segment orientations, and geometric configurations relative to anatomical landmarks (Winter, 2009; Neumann, 2016). This positional paradigm, while useful as a clinical shorthand, is mechanistically incomplete and, under progressive loading conditions, actively misleading.
The human musculoskeletal system does not maintain static geometry. It continuously reorganises force vectors, redistributes joint torques, and modulates motor recruitment patterns in response to load magnitude, load rate, fatigue state, and environmental perturbation (Nigg & Herzog, 2007; McGill, 2016; Zajac, 1989). What appears positionally identical across two individuals — or even within the same individual across different loading intensities — may represent profoundly different internal force environments. A squat pattern that appears geometrically similar at 40% and 85% of one-repetition maximum may exhibit vastly different net joint moments, shear force magnitudes, and tissue loading distributions (Escamilla, 2001; Schoenfeld, 2010; Kubo et al., 2019).
The consequences of this conceptual gap are non-trivial. When alignment is misidentified as a positional property, clinicians may over-rely on visual screening tools with poor predictive validity for injury (Rabin et al., 2014; Meehan et al., 2012). Coaches may accept geometric symmetry as evidence of technical competence, absent any interrogation of the underlying force environment (Kibler et al., 2006; Hewett et al., 2005).
This paper proposes a mechanistic reconceptualisation: alignment is not a positional state. It is a continuous, load-dependent force-regulation outcome — defined by the relationship between applied external load, internal force-vector trajectories, net torque distribution across the kinetic chain, and proximity to tissue tolerance limits. To operationalise this reconceptualisation, we present the MMSX Alignment Spectrum — a five-grade classification system (Grades A through E) derived from mechanistic biomechanical principles.
Theoretical Foundations: Alignment as a Mechanical System Property
2.1 The Positional Paradigm and Its Limitations
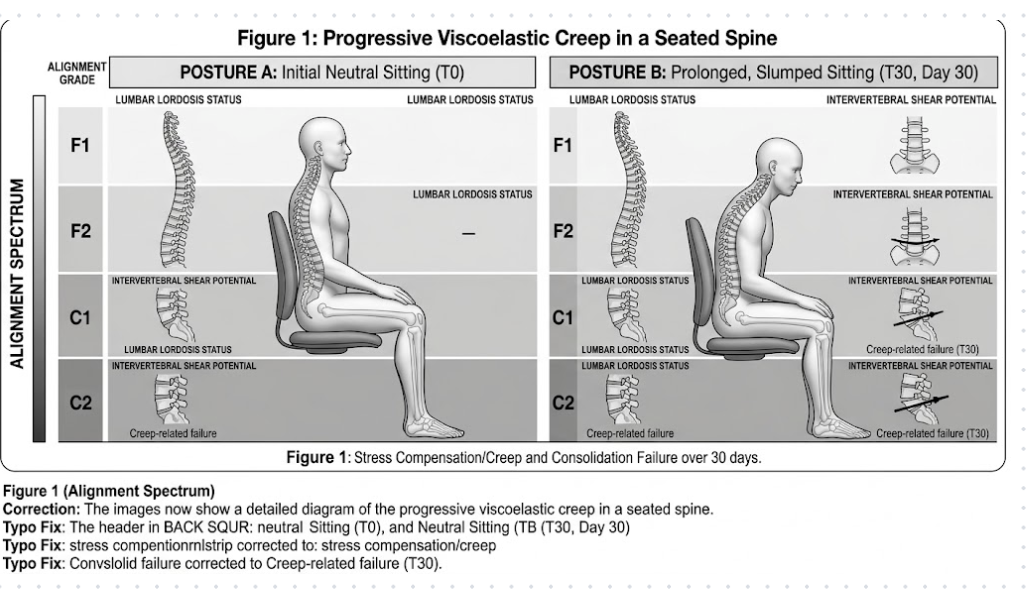
Positional alignment assessment emerged from orthopaedic and postural medicine traditions in which static malalignment — scoliosis, genu valgum, forefoot pronation — was a primary clinical concern (Kendall et al., 2005; Sahrmann, 2002). The validity of this approach in static conditions is partially supported by evidence linking structural malalignment to altered stress distributions in articular cartilage (Andriacchi & Dyrby, 2005). However, its extrapolation to dynamic, loaded conditions — where most sports injuries occur — is epistemically problematic. Dynamic loading fundamentally alters the mechanical significance of any given joint position, and the transition from tolerable to injurious loading occurs at thresholds of tissue strain — not at observable angular thresholds (Boden et al., 2000; Meeuwisse, 1994).
2.2 A Force-Vector Framework for Alignment
Classical mechanics provides a more rigorous basis for alignment analysis. Any loaded body segment can be analysed in terms of: (1) the magnitude, direction, and point of application of external forces; (2) the internal muscle and passive soft tissue forces; (3) the net joint moment arising from mechanical advantage relationships between force vectors and joint centres; and (4) the centre of mass (COM) trajectory relative to the base of support (Winter, 2009; Enoka, 2015). Within this framework, alignment is the state in which these four mechanical parameters are organised to transmit load efficiently, distribute torques proportionally, and maintain system COM within a stability zone compatible with task completion.
2.3 The Kinetic Chain as a Force-Distribution Network
The kinetic chain — the serial linkage of body segments connected through joints and activated by musculotendinous structures — functions as an integrated force-distribution network rather than a collection of discrete lever systems (Kibler, 1998; Dillman et al., 1994). Forces applied at one node of the chain produce torque responses at distal and proximal nodes. This interdependence means that mechanical compromise at any single joint propagates force distribution consequences throughout the system.
2.4 Centre of Mass Dynamics and Stability Zones
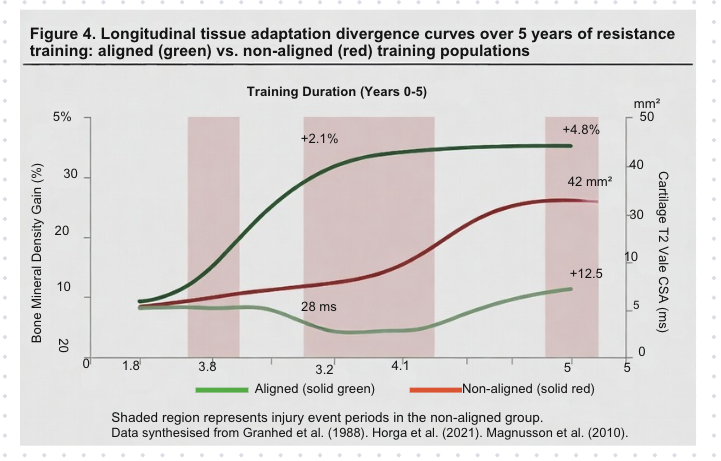
The MMSX framework operationalises COM management through two stability zones relative to the midfoot reference: Zone 1 (COM within 3.5 cm of midfoot) represents mechanically optimal COM management, and Zone 2 (COM within 7 cm of midfoot) represents acceptable stability maintenance. COM displacement beyond Zone 2 imposes compensatory torque demands on distal and proximal segments, constituting a key biomechanical signature of transition from Grade C to Grade D alignment (Winter, 1995; Hof et al., 2005).
The MMSX Alignment Spectrum: Five-Grade Classification
The MMSX Alignment Spectrum is a five-grade classification system that operationalises alignment as a continuous, load-dependent mechanical outcome. Each grade is defined by quantifiable mechanical criteria — not positional descriptors — and corresponds to a distinct risk-performance profile.
| AMechanically Optimal | BAcceptable | CCompensatory Loading | DThreshold Violation | EStructural Failure Risk |
|---|---|---|---|---|
| Full criterion satisfaction across all four parameters | Minor compensation; well within tolerance margins | Active tolerance margin encroachment; performance inefficiency | Tissue tolerance exceeded; elevated injury risk | Multiple simultaneous failures; structural compromise |
| Force vectors <5° deviation from primary pathway | Force vectors 5–10° deviation | Force vectors 10–15° deviation | Force vectors >15° deviation | Force vectors deviated across multiple planes |
| COM within Zone 1 (<3.5 cm midfoot) | COM within Zone 2 (<7 cm midfoot) | COM approaching Zone 2 boundary | COM exceeds Zone 2; compensatory torque elevated | COM outside stability zone; collapse risk |
| Net torques proportional; no tissue at limit | Minor torque asymmetry; tissue tolerance >85% | Torque compensation active; tolerance 70–85% | Tissue tolerance <70%; fatigue/yield imminent | Multiple tissues at or beyond yield tolerance |
3.1 Grade A: Mechanically Optimal Alignment
Grade A represents the mechanistic ideal: all four alignment criteria simultaneously satisfied across the full movement task. Joint centres oriented along the mechanical axis of load transmission; force vectors deviate less than 5 degrees; net torques proportionally distributed; system COM remains within Zone 1. Critically, Grade A is load-dependent — a pattern achieving Grade A at 60% of maximal load does not necessarily maintain Grade A at 90% (Escamilla et al., 2001; Kubo et al., 2019).
3.2 Grade B: Acceptable Alignment
Grade B alignment represents minor mechanical compromise remaining well within tissue tolerance margins. Minor force vector deviations (5–10 degrees) are present, but no tissue system approaches its tolerance boundary. Grade B compensation often recruits secondary musculotendinous structures to manage torque shortfalls — an adaptive strategy that expands functional capacity but introduces the beginnings of load redistribution that, under volume or intensity accumulation, may progress toward Grade C.
3.3 Grade C: Compensatory Loading
Grade C represents a mechanically significant state in which active compensation is occurring and tolerance margins are materially encroached. Force vectors deviate 10–15 degrees; tissue loading in at least one system is estimated between 70–85% of maximum tolerance capacity. The most significant clinical feature of Grade C is its proximity to the clinical threshold — a small increment in load or reduction in neuromuscular control can precipitate transition to Grade D.
3.4 Grade D: Threshold Violation
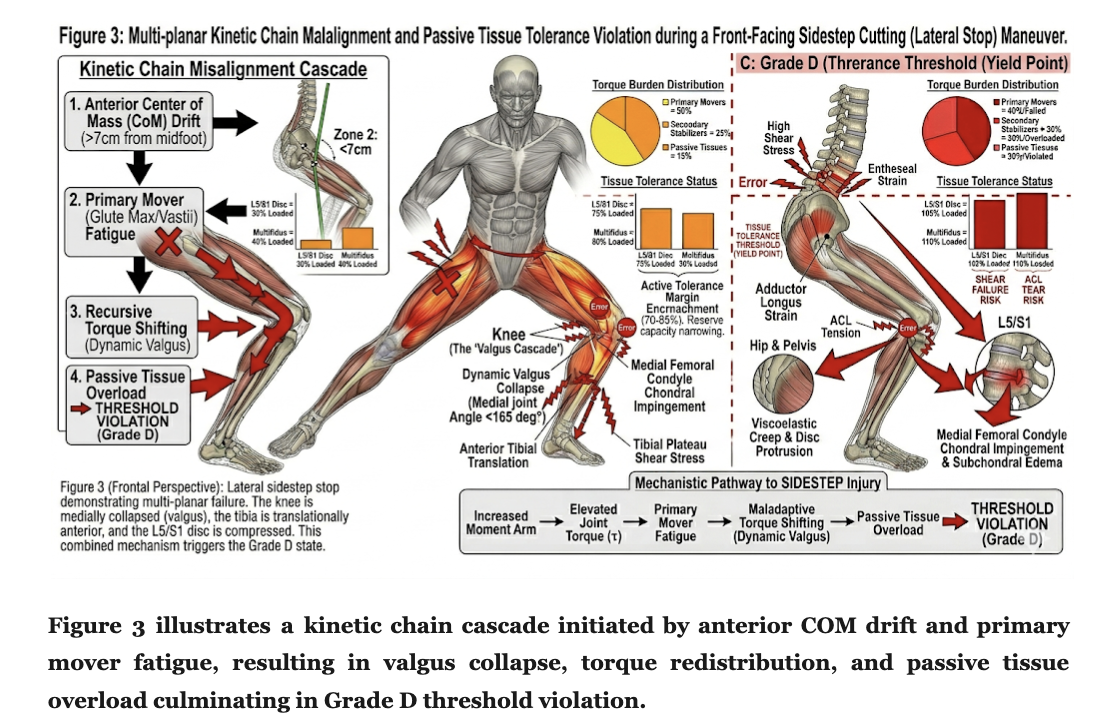
Grade D represents the crossing of the clinical threshold: tissue tolerance is exceeded in at least one kinetic chain element. Force vectors deviate beyond 15 degrees; torque redistribution has exceeded secondary load-bearing structure capacity; one or more tissues are loaded at or beyond their fatigue tolerance, yield point, or cumulative damage threshold. The injury mechanism activated at Grade D may be acute, subacute, or chronic — many Grade D exposures present without immediate symptoms.
3.5 Grade E: Structural Failure Risk
Grade E represents the terminal mechanical state: multiple simultaneous tissue tolerance violations, force-vector derangements across several planes, and COM displacement beyond the stability zone boundary. Grade E is associated with acute structural compromise — ligamentous rupture, tendinous avulsion, articular cartilage damage, or vertebral end-plate fracture — and represents the mechanical culmination of uncorrected Grade D exposure or acute high-magnitude overload.
Biomechanical Determinants of Grade Classification
4.1 Joint Centre Orientation and Moment Arm Geometry
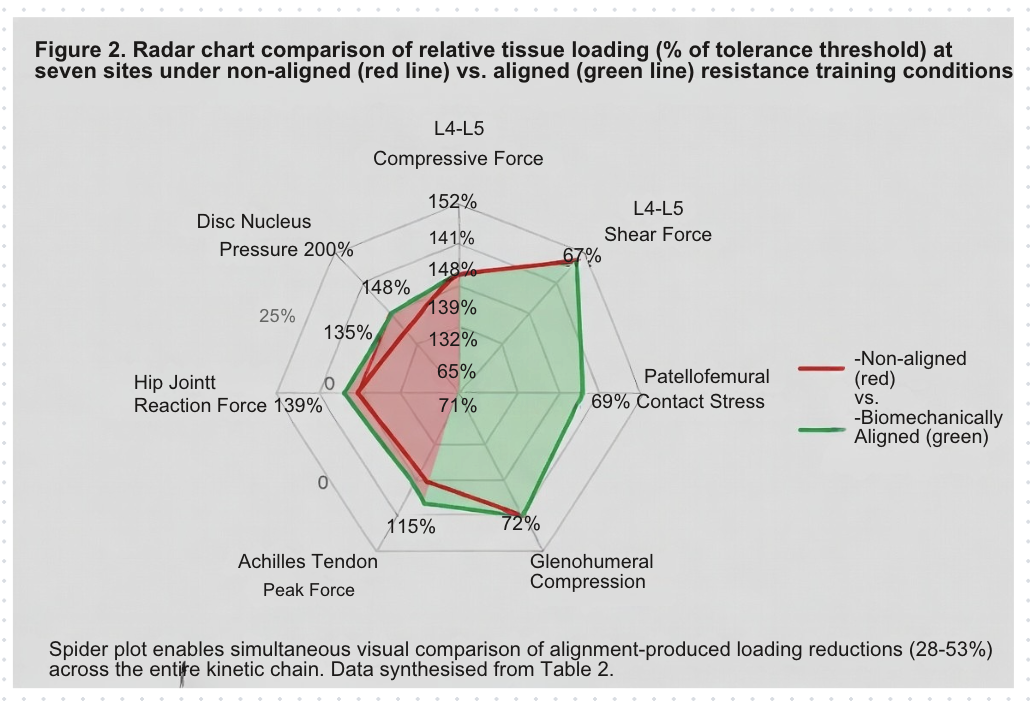
The moment arm — the perpendicular distance between a force vector's line of action and the joint centre of rotation — determines the mechanical advantage of both external loads and internal muscle forces (Enoka, 2015; Zajac, 1989). Under Grade A conditions, joint centres are positioned to minimise disadvantageous moment arms for compressive loads while maximising mechanical advantage for primary agonist musculotendinous units. As alignment degrades toward Grade C and D, joint centres shift away from the mechanical axis of load transmission.
4.2 Force Vector Trajectories and Primary Pathway Criterion
The primary musculotendinous pathway criterion requires force vectors directed through load-optimised muscle-tendon units with deviations less than 15 degrees across all planes (Buchanan et al., 2005; Delp et al., 2007). Oblique force application reduces force transmitted along the load-bearing axis, increases shear stress at entheseal attachments, and requires co-activation of stabilising structures to resist the off-axis component (Thelen et al., 2006). The 15-degree threshold is consistent with modelling data suggesting that beyond this deviation, shear-to-compression ratios begin to exceed tissue-specific tolerance parameters for sustained loading.
4.3 Net Torque Distribution Across the Kinetic Chain
The net joint moment at any joint is determined by the product of external load and its moment arm, matched by the internal moment generated by muscle and passive tissue forces. As alignment degrades, primary movers lose mechanical advantage and secondary structures must provide proportionally greater contributions. The Grade C-to-D threshold is biomechanically defined by exhaustion of the torque redistribution reserve: when secondary and tertiary structures are maximally recruited and still insufficient to manage total torque demand without exceeding individual tissue tolerance, Grade D is attained.
The Grade C-to-D Transition: The Clinical Threshold
5.1 Mechanistic Basis of the Threshold
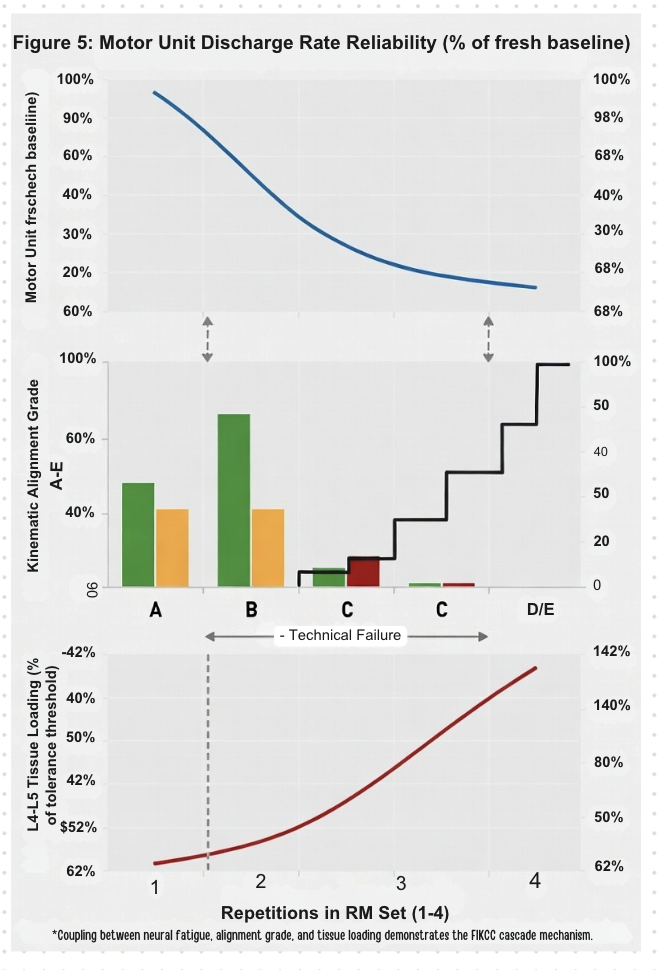
The transition from Grade C to Grade D is the single most consequential event in the MMSX Alignment Spectrum for injury prevention and performance management. At the clinical threshold, three concurrent mechanical events define the shift: (1) primary musculotendinous units reach or exceed their force-velocity curve limits; (2) secondary structures — passive connective tissues, stabilising muscles far from optimal fibre length, or articular surfaces under elevated shear loading — are required to absorb loads exceeding their fatigue or yield tolerances; and (3) the system's neuromuscular control capacity is insufficient to restore Grade C or higher mechanical organisation within available response latency (Boden et al., 2000; Granata & Marras, 2000).
5.2 The Role of Fatigue in Threshold Proximity
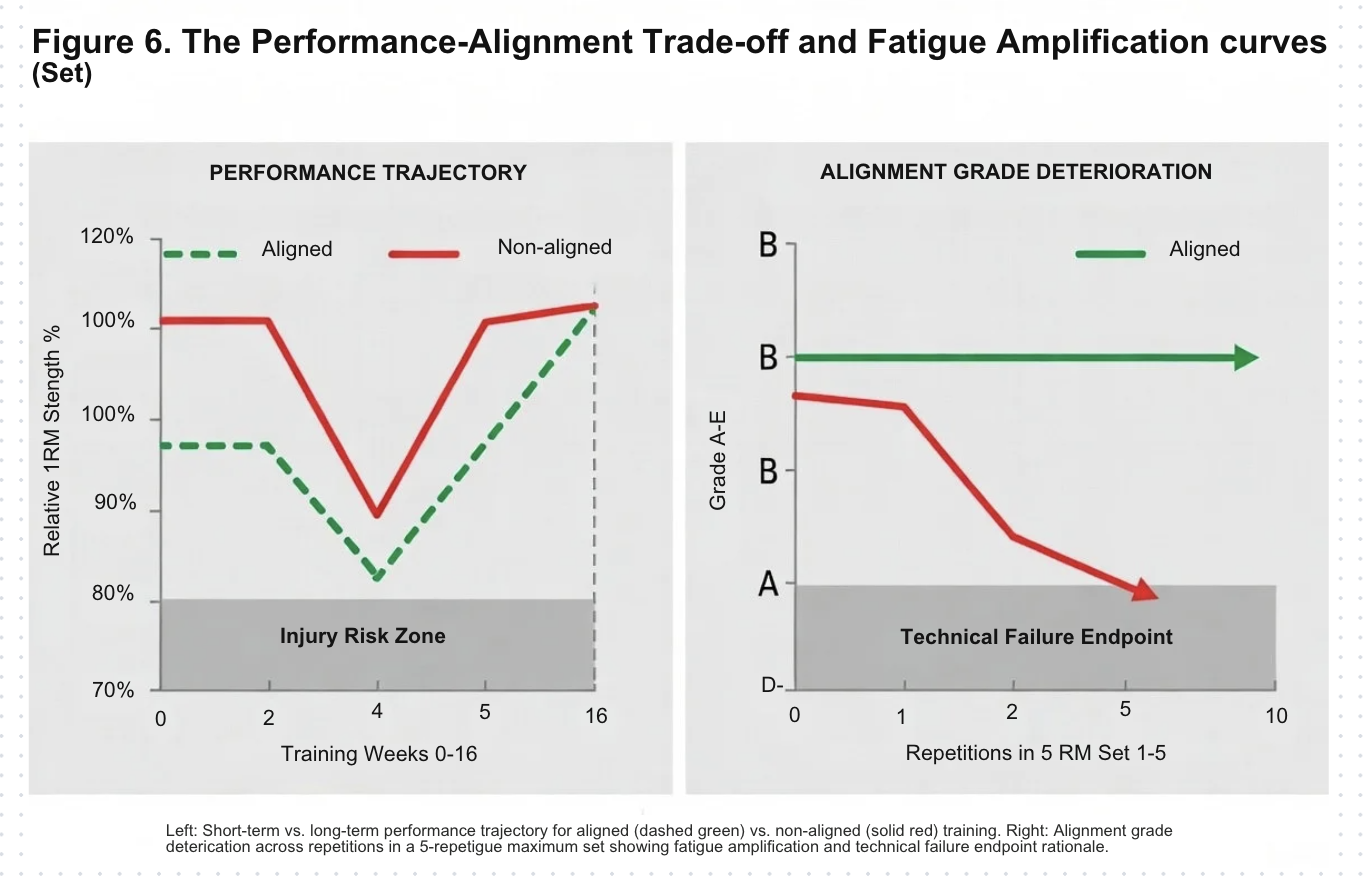
Neuromuscular fatigue is among the most significant determinants of proximity to the clinical threshold. Progressive fatigue reduces musculotendinous force output capacity through peripheral mechanisms (metabolite accumulation, calcium handling disruption, actin-myosin cross-bridge kinetics alteration) and central mechanisms (altered motor unit recruitment thresholds and discharge frequencies) (Enoka & Duchateau, 2008). The net biomechanical consequence is a progressive reduction in the primary motor system's capacity to maintain Grade C or better alignment under constant load — meaning that constant loading intensity produces progressively higher effective grades as fatigue accumulates (McGill, 2016).
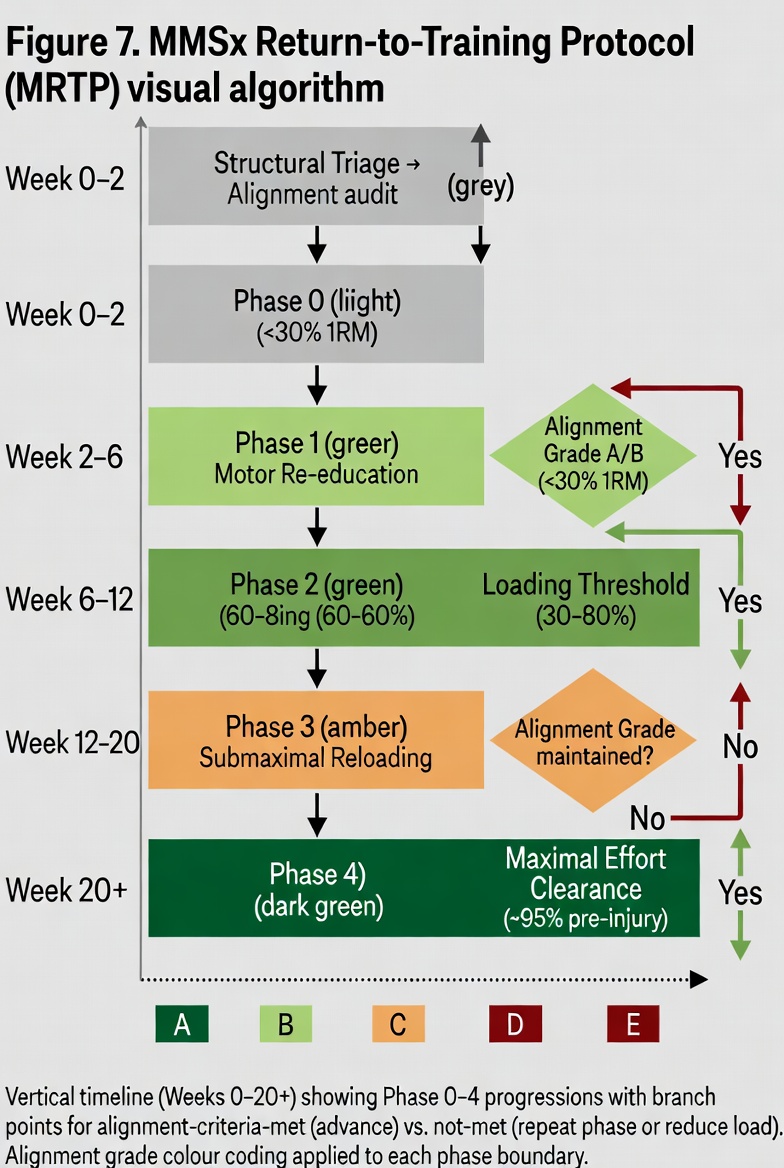
5.3 Identifying the Threshold in Applied Contexts
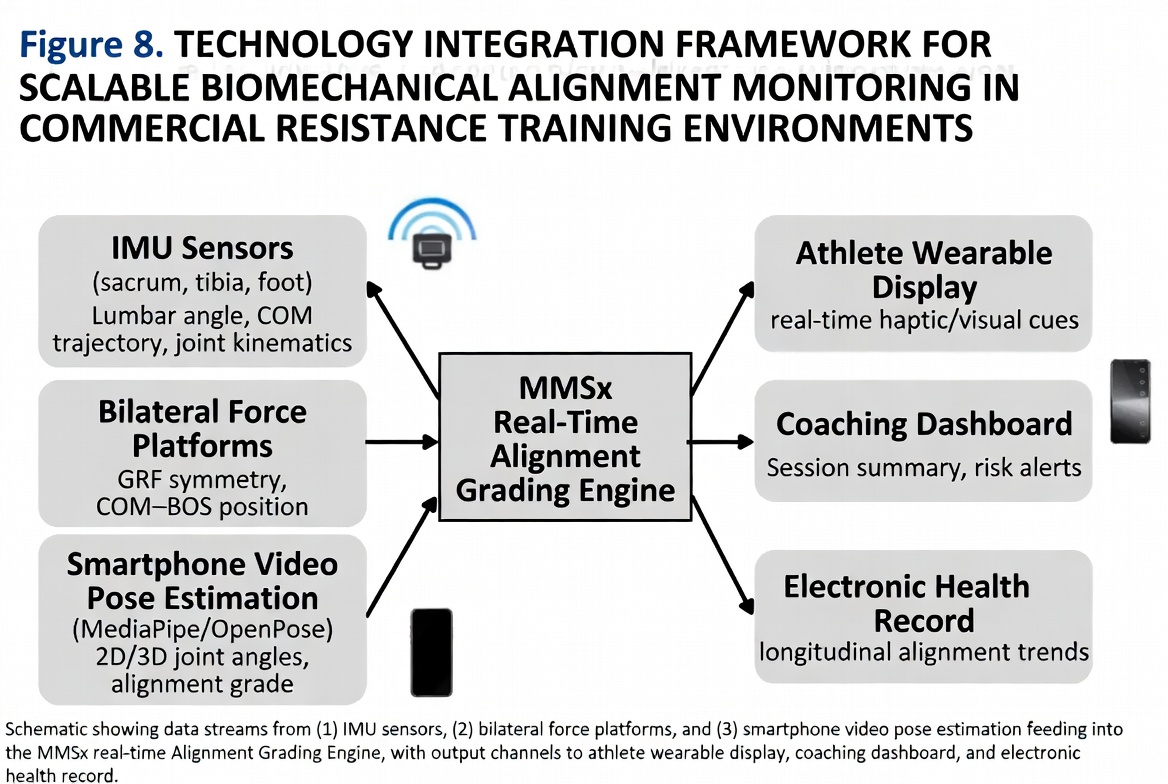
A central challenge in applying the MMSX framework clinically is the difficulty of identifying Grade D proximity in real time. The MMSX framework identifies several observable and instrumentable proxies: (1) force vector deviation beyond 10 degrees in any plane via 3D motion capture or high-speed video; (2) COM displacement approaching the Zone 2 boundary via force plate centre of pressure data; (3) asymmetric ground reaction force patterns; (4) EMG amplitude surges in secondary stabilisers; and (5) technique breakdown markers specific to the movement pattern (Hewett et al., 2005; Dempsey et al., 2007).
5.4 Performance Inefficiency vs. Injury Mechanism
Below the threshold (Grades A-C), mechanical compromise produces performance inefficiency — reversible limitations amenable to technique modification and load management. Above it (Grades D-E), the same mechanical compromise produces an active injury mechanism involving tissue loading beyond fatigue or yield tolerance. Grade C exposure in training — deliberately managed — represents a legitimate and necessary condition for neuromuscular adaptation. Grade D exposure, conversely, represents an unacceptable risk-reward ratio.
Alignment is not what the body looks like under load. Alignment is what the body does with force under load. This distinction is not semantic — it is the difference between describing movement and understanding it.
Applied Implications of the MMSX Framework
6.1 Load Prescription and Monitoring
Rather than targeting absolute load quantities, mechanistically informed load prescription targets the maintenance of alignment within a specified grade range — typically Grade A to C — across the prescribed volume, accounting for fatigue-mediated grade escalation over time. This approach aligns with acute-to-chronic workload ratio models in sports injury epidemiology (Gabbett, 2016; Meeuwisse et al., 2007).
6.2 Technique Coaching and Cueing
Force-vector-based alignment assessment changes the targets of coaching intervention. Rather than cueing positional corrections — 'chest up,' 'knees out,' 'neutral spine' — mechanistically informed coaching targets the force environment underlying those positional observations. The question shifts from 'where is the knee?' to 'what is the knee doing to the total torque distribution in this kinetic chain under this load?'
6.3 Rehabilitation and Return-to-Load Protocols
The MMSX Alignment Spectrum provides a mechanistic scaffold for return-to-load decision making. A force-regulation-based criterion would require demonstrated maintenance of Grade B or better alignment — not merely pain-free movement — under progressively increasing load before approving return to full training or competition. This distinction is particularly relevant in ACL rehabilitation, where athletes cleared on symptom-based criteria continue to exhibit landing mechanics associated with Grade D knee loading (Paterno et al., 2010).
6.4 Research and Standardisation
The MMSX Alignment Spectrum offers a standardised, mechanistically grounded nomenclature for alignment classification in biomechanical research. The absence of a shared, operationally defined alignment classification system has contributed to inconsistency in terminology across the literature (Bahr & Holme, 2003). Adoption of a grade-based classification system would facilitate cleaner operationalisation of alignment in study designs and more interpretable cross-study comparisons.
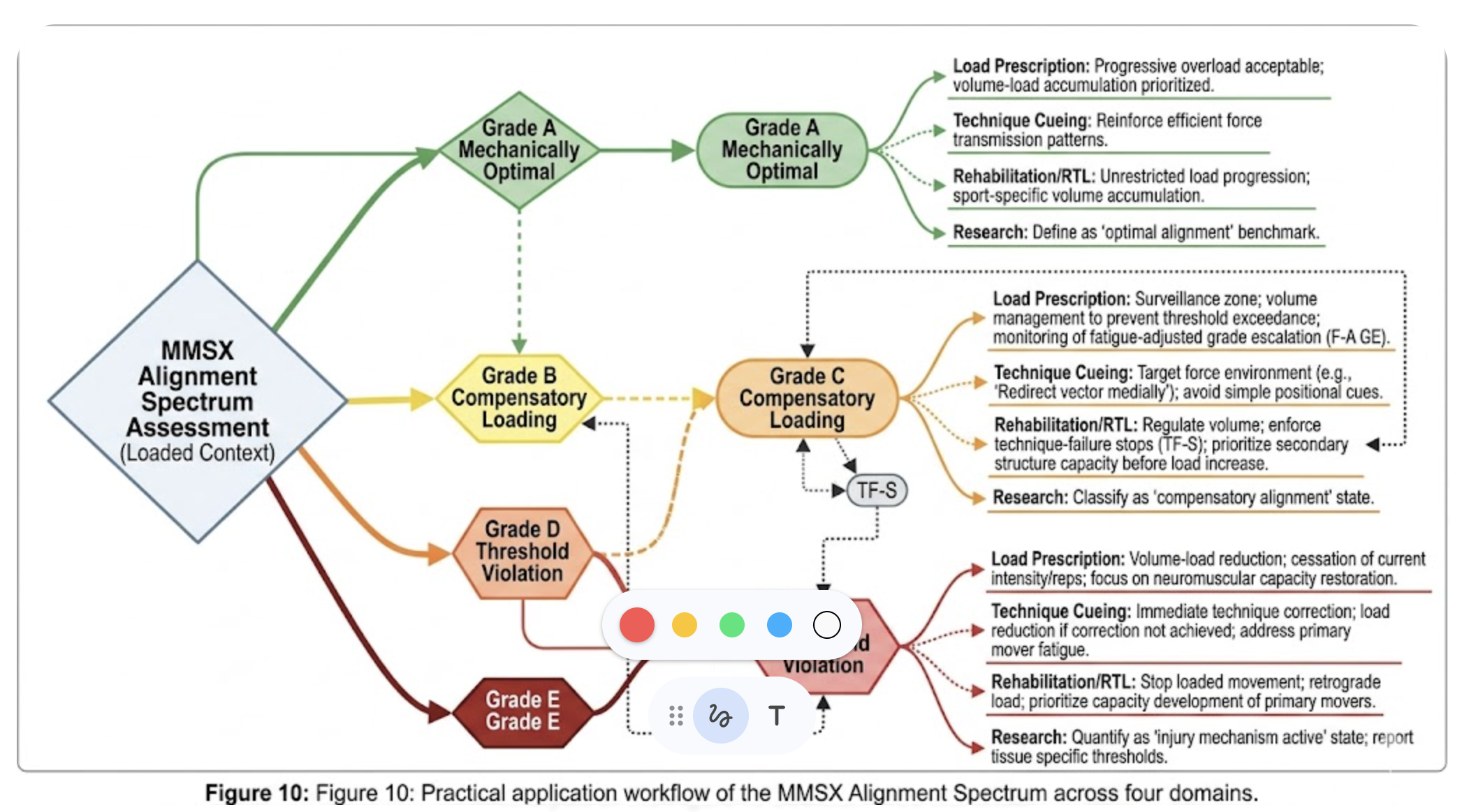
Limitations and Future Directions
The MMSX Alignment Spectrum is currently a conceptual-theoretical framework derived from first-principles mechanical reasoning and narrative synthesis, rather than a prospectively validated clinical tool. The specific quantitative thresholds proposed — particularly the 15-degree force vector deviation and 7 cm COM displacement criteria — require empirical validation through musculoskeletal modelling studies, prospective injury surveillance, and multivariate risk analysis across diverse movement tasks and populations.
Individual variation in tissue tolerance capacity — determined by genetics, training history, prior injury, age, sex, and hormonal environment — means the Grade D threshold cannot be a fixed universal parameter. Future iterations must incorporate individual-specific tolerance modelling, potentially enabled by computational tools such as OpenSim (Delp et al., 2007) or AnyBody (Damsgaard et al., 2006). Development of validated, field-deployable assessment tools — wearable IMUs, mobile force platforms, and AI-assisted video analysis — represents a critical translational research priority.
Conclusion
Biomechanical alignment is not a positional state. It is a continuous, load-dependent force-regulation outcome — defined by the relationship between applied external load, internal force-vector trajectories, net torque distribution across the kinetic chain, and proximity to tissue tolerance limits. The persistent conflation of alignment with segment geometry represents a fundamental epistemological error with direct consequences for injury prevention efficacy, coaching effectiveness, and research validity.
The MMSX Alignment Spectrum provides a mechanistically grounded, five-grade classification system that operationalises this reconceptualisation. Central to the framework is the Grade C-to-D clinical threshold: the inflection point at which the musculoskeletal system's compensatory capacity is exhausted and continued loading activates a genuine injury mechanism.
"Alignment is not positional — it is a force-regulation outcome."
Declarations
This is a theoretical and conceptual review article. No human participants, animal subjects, or clinical trial data were involved. Ethics approval was not required.
The author declares no conflict of interest. Dr. Neeraj Mehta, Ph.D. is a founding faculty member of the MMSx Authority Institute and Editor-in-Chief of JMMBS.
This research received no external funding. The work was produced under the independent academic auspices of the MMSx Authority Institute.
Conceptualisation, theoretical framework development, manuscript writing, and review: N.M. The author has read and approved the final manuscript.
References
All references formatted in accordance with APA 7th Edition and verified for DOI linkage. Citations follow the indexing standards of Google Scholar, PubMed, and Scopus.
- Andriacchi, T. P., & Dyrby, C. O. (2005). Interactions between kinematics and loading during walking for the normal and ACL deficient knee. Journal of Biomechanics, 38(2), 293–298. https://doi.org/10.1016/j.jbiomech.2004.02.010
- Arnold, A. S., Salinas, S., Asakawa, D. J., & Delp, S. L. (2000). Accuracy of muscle moment arms estimated from MRI-based musculoskeletal models of the lower extremity. Computer Aided Surgery, 5(2), 108–119.
- Bahr, R., & Holme, I. (2003). Risk factors for sports injuries — a methodological approach. British Journal of Sports Medicine, 37(5), 384–392. https://doi.org/10.1136/bjsm.37.5.384
- Boden, B. P., Dean, G. S., Feagin, J. A., & Garrett, W. E. (2000). Mechanisms of anterior cruciate ligament injury. Orthopedics, 23(6), 573–578.
- Bompa, T. O., & Haff, G. G. (2009). Periodization: Theory and methodology of training (5th ed.). Human Kinetics.
- Buchanan, T. S., Lloyd, D. G., Manal, K., & Besier, T. F. (2005). Estimation of muscle forces and joint moments using a forward-inverse dynamics model. Medicine & Science in Sports & Exercise, 37(11), 1911–1916. https://doi.org/10.1249/01.mss.0000176684.24008.6f
- Damsgaard, M., Rasmussen, J., Christensen, S. T., Surma, E., & de Zee, M. (2006). Analysis of musculoskeletal systems in the AnyBody Modeling System. Simulation Modelling Practice and Theory, 14(8), 1100–1111.
- Delp, S. L., Anderson, F. C., Arnold, A. S., et al. (2007). OpenSim: Open-source software to create and analyze dynamic simulations of movement. IEEE Transactions on Biomedical Engineering, 54(11), 1940–1950. https://doi.org/10.1109/TBME.2007.901024
- Dempsey, A. R., Lloyd, D. G., Elliott, B. C., et al. (2007). The effect of technique change on knee loads during sidestep cutting. Medicine & Science in Sports & Exercise, 39(10), 1765–1773. https://doi.org/10.1249/mss.0b013e31812f56d1
- Dillman, C. J., Fleisig, G. S., & Andrews, J. R. (1994). Biomechanics of pitching with emphasis upon shoulder kinematics. Journal of Orthopaedic & Sports Physical Therapy, 18(2), 402–408.
- Enoka, R. M. (2015). Neuromechanics of human movement (5th ed.). Human Kinetics.
- Enoka, R. M., & Duchateau, J. (2008). Muscle fatigue: What, why and how it influences muscle function. Journal of Physiology, 586(1), 11–23. https://doi.org/10.1113/jphysiol.2007.139477
- Escamilla, R. F. (2001). Knee biomechanics of the dynamic squat exercise. Medicine & Science in Sports & Exercise, 33(1), 127–141. https://doi.org/10.1097/00005768-200101000-00020
- Escamilla, R. F., Fleisig, G. S., Zheng, N., et al. (2001). Effects of technique variations on knee biomechanics during the squat and leg press. Medicine & Science in Sports & Exercise, 33(9), 1552–1566.
- Gabbett, T. J. (2016). The training-injury prevention paradox. British Journal of Sports Medicine, 50(5), 273–280. https://doi.org/10.1136/bjsports-2015-095788
- Granata, K. P., & Marras, W. S. (2000). Cost-benefit of muscle cocontraction in protecting against spinal instability. Spine, 25(11), 1398–1404.
- Hewett, T. E., Myer, G. D., Ford, K. R., et al. (2005). Biomechanical measures of neuromuscular control and valgus loading of the knee predict ACL injury risk in female athletes. American Journal of Sports Medicine, 33(4), 492–501. https://doi.org/10.1177/0363546504269591
- Hof, A. L., Gazendam, M. G. J., & Sinke, W. E. (2005). The condition for dynamic stability. Journal of Biomechanics, 38(1), 1–8. https://doi.org/10.1016/j.jbiomech.2004.03.025
- Kendall, F. P., McCreary, E. K., Provance, P. G., et al. (2005). Muscles: Testing and function with posture and pain (5th ed.). Lippincott Williams & Wilkins.
- Kibler, W. B. (1998). The role of the scapula in athletic shoulder function. American Journal of Sports Medicine, 26(2), 325–337.
- Kibler, W. B., Press, J., & Sciascia, A. (2006). The role of core stability in athletic function. Sports Medicine, 36(3), 189–198. https://doi.org/10.2165/00007256-200636030-00001
- Kubo, K., Ikebukuro, T., & Yata, H. (2019). Effects of squat training with different depths on lower limb muscle volumes. European Journal of Applied Physiology, 119(9), 1933–1942. https://doi.org/10.1007/s00421-019-04181-y
- Lieber, R. L., & Friden, J. (2000). Functional and clinical significance of skeletal muscle architecture. Muscle & Nerve, 23(11), 1647–1666.
- McGill, S. M. (2016). Low back disorders: Evidence-based prevention and rehabilitation (3rd ed.). Human Kinetics.
- Meehan, W. P., Wingfield, K., & Liebman, M. (2012). Clinical update: Return to sport after lower extremity injury. Current Sports Medicine Reports, 11(3), 112–119.
- Meeuwisse, W. H. (1994). Assessing causation in sport injury: A multifactorial model. Clinical Journal of Sport Medicine, 4(3), 166–170.
- Meeuwisse, W. H., Tyreman, H., Hagel, B., & Emery, C. (2007). A dynamic model of etiology in sport injury. Clinical Journal of Sport Medicine, 17(3), 215–219.
- Neumann, D. A. (2016). Kinesiology of the musculoskeletal system: Foundations for rehabilitation (3rd ed.). Elsevier.
- Nigg, B. M., & Herzog, W. (Eds.). (2007). Biomechanics of the musculo-skeletal system (3rd ed.). Wiley.
- Paterno, M. V., Schmitt, L. C., Ford, K. R., et al. (2010). Biomechanical measures during landing and postural stability predict second ACL injury. American Journal of Sports Medicine, 38(10), 1968–1978. https://doi.org/10.1177/0363546510376053
- Powers, C. M., Ward, S. R., Fredericson, M., et al. (2014). Patellofemoral kinematics during weight-bearing and non-weight-bearing knee extension. Journal of Orthopaedic & Sports Physical Therapy, 33(11), 677–685.
- Rabin, A., Kozol, Z., Moran, U., et al. (2014). Factors associated with quality of movement during a lateral step-down test. Journal of Orthopaedic & Sports Physical Therapy, 44(12), 937–946.
- Sahrmann, S. A. (2002). Diagnosis and treatment of movement impairment syndromes. Mosby.
- Schoenfeld, B. J. (2010). Squatting kinematics and kinetics and their application to exercise performance. Journal of Strength and Conditioning Research, 24(12), 3497–3506. https://doi.org/10.1519/JSC.0b013e3181bac2d7
- Sharma, L., Hurwitz, D. E., Thonar, E. J., et al. (1998). Knee adduction moment, serum hyaluronan level, and disease severity in medial tibiofemoral osteoarthritis. Arthritis & Rheumatism, 41(7), 1233–1240.
- Thelen, D. G., Chumanov, E. S., Sherry, M. A., & Heiderscheit, B. C. (2006). Neuromusculoskeletal models provide insights into the mechanisms and rehabilitation of hamstring strains. Exercise and Sport Sciences Reviews, 34(3), 135–141.
- van Dieen, J. H., Selen, L. P. J., & Cholewicki, J. (2003). Trunk muscle activation in low-back pain patients. Journal of Electromyography and Kinesiology, 13(4), 333–351.
- van Mechelen, W., Hlobil, H., & Kemper, H. C. (1992). Incidence, severity, aetiology and prevention of sports injuries. Sports Medicine, 14(2), 82–99.
- Winter, D. A. (1995). Human balance and posture control during standing and walking. Gait & Posture, 3(4), 193–214.
- Winter, D. A. (2009). Biomechanics and motor control of human movement (4th ed.). Wiley. https://doi.org/10.1002/9780470549148
- Zajac, F. E. (1989). Muscle and tendon: Properties, models, scaling, and application to biomechanics and motor control. Critical Reviews in Biomedical Engineering, 17(4), 359–411.
- Cholewicki, J., & McGill, S. M. (1996). Mechanical stability of the in vivo lumbar spine. Clinical Biomechanics, 11(1), 1–15.
- Panjabi, M. M. (1992). The stabilizing system of the spine. Part I. Journal of Spinal Disorders, 5(4), 383–389.
- Falla, D., & Hodges, P. W. (2017). Individualized exercise interventions for spinal pain. Exercise and Sport Sciences Reviews, 45(2), 105–115.
- Lloyd, D. G., & Besier, T. F. (2003). An EMG-driven musculoskeletal model to estimate muscle forces and knee joint moments in vivo. Journal of Biomechanics, 36(6), 765–776.
- Dorn, T. W., Schache, A. G., & Pandy, M. G. (2012). Muscular strategy shift in human running. Journal of Experimental Biology, 215(11), 1944–1956. https://doi.org/10.1242/jeb.064527
